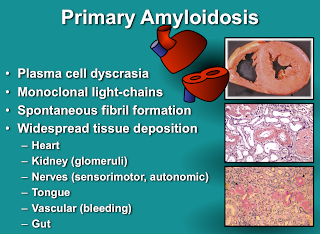
Any organ can be affected, but heart involvement in particular drives survival. Generally, the more heart-involvement by amyloid, the worse the outcome. This is the basis of the cardiac staging system for AL amyloidosis developed by Dr. Angela Dispenzieri and her colleagues at the Mayo Clinic. The original version of this staging system used simple blood tests (NT-pro-BNP, and either cTnT or cTnI) to divide newly-diagnosed patients with AL amyloidosis into three groups:
 |
| Staging system using cardiac biomarkers to predict survival of patients with AL amyloidosis (ref: http://jco.ascopubs.org/content/22/18/3751.long) |
An updated version of this staging system incorporates another blood test, the serum free light chain measurement (FreeLite Test), resulting in 4 different stages.
When the heart is filled with amyloid, it becomes thick and stiff. The thickness can be measured using echocardiography (an "ECHO"). The affected heart often doesn't relax normally after contracting ("diastolic dysfunction"). This can lead to congestive heart failure. The electrical conduction system of the heart may become compromised, and patients may be at risk for life threatening heart rhythm abnormalities or cardiac arrest. This latter problem is the explanation for the steep drop in survival in advanced stage patients the first year after diagnosis, and preventing arrhythmias can be one of the keys to survival.
It is widely held that the heart dysfunction in amyloidosis is the result of amyloid infiltrating the heart tissue - like impregnating the tissue with wax or concrete, making it impossible for the heart muscle cells (myocardiocytes) to contract, and disrupting the electrical circuitry of the organ.
In medical school I imagined it like the La Brea Tar Pits - a sticky, stiff mire that eventually exhausted any living thing that got stuck in it. My daughter suggested that Frodo enmeshed in Shelob's web would have been a cooler analogy. We debated this for a while, each of us conveniently ignoring our gnawing concerns that geekiness is potentially inheritable.
In medical school I imagined it like the La Brea Tar Pits - a sticky, stiff mire that eventually exhausted any living thing that got stuck in it. My daughter suggested that Frodo enmeshed in Shelob's web would have been a cooler analogy. We debated this for a while, each of us conveniently ignoring our gnawing concerns that geekiness is potentially inheritable.
 |
| Prehistoric elephant in tar pit, doing an impression of a cardiac muscle cell in an amyloid-filled heart. Low art, even by Neanderthal standards. (Image ref: http://www.freeimageslive.co.uk/files/images006/mammouth_tar_pits.jpg) |
At any rate, the actual story is a bit more complicated. It is not only the accumulation of amyloid fibers (made up of aggregated light chains) around the heart muscle cells which cause heart dysfunction. Researchers from Boston have shown that AL light chains themselves (ones that have not been incorporated into fibrils) are directly toxic to myocardiocytes. They reduce the ability of the heart muscle cells to contract, and eventually they can cause the cells to die. This seems to be caused by activation of a signaling protein called p38-alpha MAPK inside the cells (click here to see the original article). Of interest, normal non-amyloid-forming light chains - ones made by normal, non-clonal plasma cells - do not damage heart muscle cells like this. Of even more interest is that various pharmaceutical companies are developing and testing p38-alpha MAPK inhibitors. In theory, such agents could minimize the direct toxic effects of AL-light chains on the heart, and maybe - just maybe - improve the prospects of patients with advanced cardiac AL amyloidosis.