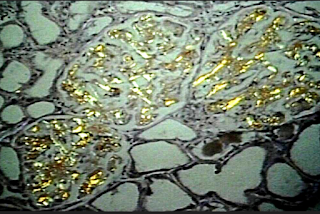
Tomoaki Murakami and colleagues describe their research related to an outbreak of AA amyloidosis occurring at a Japanese poultry farm. Avian AA amyloidosis is well characterized (as indicated by its inclusion in Table II from the most recent amyloid fibril nomenclature committee guidelines). AA amyloidosis has been described in waterfowl (due to chronic inflammatory conditions) and chickens (due to bacterial infections or repeated vaccinations). AA amyloid has also been detected in the livers of force-fed ducks and geese used for foie gras.
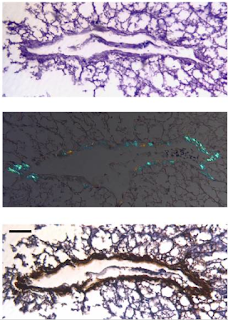 |
| A photomicrograph of a hepatic venule from a sample of commercially-available foie gras (top panel, h/e stain; middle, congo red stain under polarized light; bottom IHC confirming AA amyloid). Ref: http://www.ncbi.nlm.nih.gov/pubmed/17578924 |
The findings:
- 29 out of 38 chickens (all breeds studied) who got both vaccine SE(a) and a dose of AA fibrils (either orally or intravenously) developed AA amyloidosis. If the AA fibrils were administered orally, then the amyloidosis tended to develop in the chickens' spleens; the chickens who got the AA fibrils intravenously developed more widespread systemic amyloidosis (including the birds' intestines).
- ZERO out of 113 other chickens developed systemic AA amyloidosis.
- Serum AA precursor protein levels (SAA levels) did not correlate with risk of developing systemic AA amyloidosis. The SE(a) vaccine did not induce an increase in SAA in any subgroup of chickens (including those who developed amyloidosis) and the MG vaccine did induce a rise in SAA (with no cases of associated amyloidosis).
The scientists (and another author providing commentary in the same issue of the journal) concluded that the outbreak of avian AA amyloidosis was likely the result of chickens ingesting amyloid-contaminated feed and/or droppings after having had industry-standard vaccinations which may have pre-disposed them to developing amyloidosis.
Not an unprecedented finding. Fecal transmission is suspected to contribute to the high incidence of systemic AA amyloidosis in captive cheetahs (like cheetahs don't have enough problems). Also, the same scientists who discovered amyloidosis in foie gras fed it to amyloid-susceptible mice and found it accelerated the rate at which systemic AA amyloidosis developed. They concluded foie gras was a potential Amyloid Enhancing Factor (AEF), and that
"It would seem prudent for children and adults with rheumatoid arthritis or other diseases who are at risk for [AA amyloidosis] to avoid foods that may be contaminated with AA fibrils."Then, citing studies such as one in which formation of a specific type of amyloidosis (AApoAII) could be accelerated in predisposed experimental mice by intravenous injection of any of several different types of human amyloid fibrils (AL, ATTR, Abeta2-MG, and others), they suggest
"...that it may be hazardous for individuals who are prone to develop other types of amyloid-associated disorders, e.g., Alzheimer's disease or type II diabetes, to consume such products."Which nags at me. I don't so much wonder about whether my father, the son of a woman who died of Alzheimer's disease, should enjoy foie gras as much as he does (though maybe I should - it's Fathers' Day). I worry a little bit about whether diet could be affecting the outcome of my patients with documented systemic amyloidosis. Is our carnivorous American diet full of AEFs? Possibly. An examination of the kidneys and other organs from 302 apparently healthy cattle slaughtered for meat revealed AA amyloidosis in 5% of the animals (take a look). The incidence was lower (0.4% - 2%) in other studies. A largely vegetarian diet has been cited as a possible explanation for the virtual absence of secondary amyloidosis in leprosy patients in India compared to the fairly high incidence amongst leprosy patients in the United States, Malaysia, Brazil, and other countries. Check out this 1965 article by Amyloid Guru Alan S. Cohen and colleagues which explores this very topic. At this time, though, there is no compelling evidence for transmission or acceleration of amyloidosis (AA or any other type) in humans from ingestion of AA-containing bovine meat or organs. Additionally, a search of the medical literature did not turn up any studies looking at the impact of diet on prognosis in AL amyloidosis. So for now, I'll just worry a little bit. Comments welcome.