Any organ's function can be compromised by amyloid deposits. The kidney is one of the most commonly affected organs, and patients with injured kidneys may have symptoms, including swelling (edema) of the legs, decreased urine output, and lightheadedness due to sudden drops in blood pressure.
In order to understand how amyloid injures the kidneys, it is helpful to understand how the kidney works.
A summary of kidney anatomy and function relevant to amyloidosis:
- The kidney is made up of a million microscopic filtration units called nephrons.
- Each nephron has a filter called the glomerulus, which filters the blood.
- Some parts of the blood, namely water and electrolytes (sodium, potassium, etc), flow through this filter into a tube system where it is processed further. The material that eventually comes out of the end of all this tubing is urine.
- Other components of the blood, like red blood cells, do not pass through the filter.
- Although under normal circumstances there is essentially no protein in our urine, it is not because proteins do not pass through the filter. Filtered protein may be reabsorbed (taken back up into the body) in the first part of the tubing system. The part of the tubing where this takes place is called the proximal tubule.
- The part of the tubing further down the line is called the distal tubule.
- All of these structures, as well as blood vessels within the kidney, are surrounded by tissue which serves as scaffolding to hold it all together. This is the interstitial space, or matrix. (Its the Jell-O holding all the little pieces of fruit in place in that dessert your mother used to make on Thanksgiving)
If you want to know more about normal kidney function, see the clear, easy-to-understand post by my friend, nephrologist Joel Topf, in his blog Precious Bodily Fluids.
Light chains can injure the kidneys in a number of ways. Like albumin, normal light chains are filtered through the glomerulus and then taken back up in the proximal tubule. The receptors along the lining of the tubule which do this are actually the same for albumin and light chains (cubilin and megalin, if you were wondering). Problems develop if the light chain levels are abnormally high or if the light chains have an abnormal tendency to form clumps of strands.
- Amyloid light chains, in addition to being filtered, form deposits in the glomerulus itself. This is because these abnormal light chains are taken up by cells within the filter called mesangial cells. Mesangial cells do not take up normal light chains. After the abnormal amyloid light chains are snagged by these cells, they get processed and deposited within the matrix of the filter in strands called fibrils. When a pathologist is looking for amyloid in a kidney biopsy, s/he applies Congo Red stain, which makes these deposits look red under normal light and green under polarized light. As amyloid accumulates in the tissue around the mesangial cells in the glomerulus, the filter is damaged. It becomes "leaky" and the amount of protein lost through the filter increases.
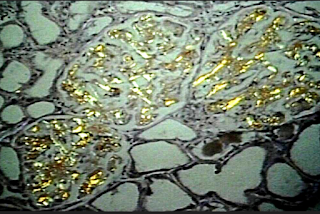 |
| Kidney biopsy stained with Congo Red stain. Top image is the view under normal light, and the bottom one is the same slide viewed under polarized light. Everything that turned fluorescent green in the second image is amyloid! Images snagged from http://www.pathguy.com/lectures/imm-iii.htm |
- Increased protein delivery to the proximal tubule is bad for one's kidneys. The receptors to reabsorb albumin and light chains can become overwhelmed. Excess albumin is lost in the urine, and it can make the urine appear "foamy." Very low blood albumin levels are what cause the swelling (edema) and low blood pressure (hypotension) amyloidosis patients experience. Even though I.V. albumin solutions exist, it is not feasible to replace it because infused albumin suffers the same fate as the patient's "home-grown" albumin: flushed. Excess light chains can also cause problems, as they can bind to other proteins in the urine and form casts (clumps that clog up the distal tubule, which in turn causes problems upstream). Cast formation is the leading cause of kidney injury in multiple myeloma, but less of an issue in amyloidosis. I posted about cast nephropathy previously (check that out).
- Inflammation plays a role in amyloid kidney injury. Abnormal light chains, when taken up by the cells in the proximal tubule actually injure the cells in that part of the kidney. Excess albumin in the tubular system and abnormal amyloid light chains in the cells lining the tubular system trigger inflammation and eventually scarring of the interstitial area. This is why AL amyloidosis patient with persisting heavy albuminuria (albumin in the urine) due to filter damage may have continued worsening of their renal function even after the amyloidosis has been treated and the light chain levels are no longer elevated: albumin-mediated kidney injury. In my own practice, this is a common and frustrating problem. One glimmer of hope: it is possible that one of the treatments commonly used in the treatment of AL amyloidosis - the proteasome inhibitor bortezomib (Velcade) - targets this inflammatory pathway. Other drugs in this same family (carfilzomib (Kyprolis) and ixazomib (MLN-9708)) are currently undergoing testing as therapy for AL amyloidosis. These drugs may not only kill bad-acting plasma cells, but also help the kidney dodge some albumin-mediated damage. Friend and colleague Meletios Dimopoulos has published extensively on this topic; check out this article describing the improvement in kidney function seen in myeloma patients who received bortezomib therapy.
Kidney transplant has been undertaken in a limited number of patients with myeloma and/or amyloidosis. A major concern is that the same disease-related processes which caused the original kidneys to fail will recur in a transplanted kidney. Also, the fact that patients with these diseases often have limited survival independent of kidney function begs the question of whether precious donor kidneys are best used in this situation. With newer therapies leading to higher remission rates and longer survival in both myeloma and AL amyloidosis, the idea that it may be time to revisit the conventional wisdom about organ transplantation is gaining traction (like here).
Lets call it a wrap. While I call this a "Patient Page," I used a lot of medical terminology. I tried to define everything in common language. Even so, it is probably clear I expect a lot from my readers. If there is anything in this page which requires clarification, TELL ME. Email me, or post it as a comment. I want the content of this (and every) post to be as clear and helpful as possible.



Hello Dr. Zonder,
ReplyDeleteWhat a fantastic explanation for anyone with kidney damage caused by Amyloidosis. I found your post to be easily understandable and very informative. I will pass this information along to the patients that attend our support group meetings. See you in Chicago this October!
Paula Schmitt
GA/FL/DC Support Group Facilitator
Amyloidosis Support Groups
www.amyloidosissupport.com
Good review. My concern is in the patients that have high 24 hr urine losses of protein (>10 gm/day) accompanied by low measured serum pre-albumin. In addition to chemotherapy, would you recommend increased dietary protein intake (assuming a normal CrCl) to compensate for the losses?
ReplyDeleteKevin Anderson, MD
(I am also an amyloidosis patient)
Kevin, thats a great question. Increasing dietary protein intake will not effectively compensate for the ongoing renal losses. Even intravenous albumin supplementation is of little benefit (certainly not sustained benefit). I welcome any input from nephrologists who might stumble across this thread.
ReplyDeleteI am a doctor and I was looking into trying to explain this difficult topic to my medical student and patients. Really good and clear review - even improved my insight! Thanks! I will refer people to this article.
ReplyDelete