Here it is:
https://storify.com/Amyloid_Planet/amyloidosisjc-vol-7-march-28th-2016#publicize
A nice discussion about a very difficult topic. Grateful to have the lead author from the paper, Dr. Laura Dember from U Penn, join us!
Paper and date for next installment will be announced soon!
28 March, 2016
26 March, 2016
#amyloidosisJC 3/28/16 @ 9 pm EST: Eprodisate for the Treatment of Renal Disease in AA #amyloidosis
This installment of #amyloidosisJC examines the not-positive-enough results of a randomized trial comparing the effects of eprodisate to placebo in AA amyloidosis patients with amyloid-related renal disease. Click HERE for a link to the actual article. I'd like once again to thank Dr. Naresh Bumma (@NB191186) for his assistance preparing this post.
 |
| Dember, Laura M., et al. "Eprodisate for thetreatment of renal disease in AA amyloidosis." New England Journal of Medicine 356.23 (2007): 2349-2360. |
Background: The amyloidoses are a group of diseases characterized by extracellular deposition of fibrils comprised of misfolded proteins and other molecules such as glycosaminoglycans and SAP. End organ damage may result from either the fibrillar deposits or toxic oligomers and/or protofilaments. AA amyloidosis is a rare entity, almost always seen in the setting of an underlying chronic inflammatory condition. The amyloid-forming protein is a proteolytic product of serum amyloid A protein (SAA), an acute phase reactant protein produced by the liver. The kidney is the most commonly affected organ, though the GI tract and other organs can also be involved.
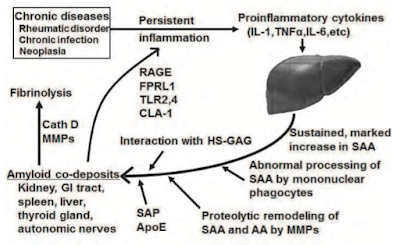 |
| AA Amyloidosis pathophysiology from T. Nakamura (click HERE for link to paper) |
Trial Design: Multicenter, randomized, double-blind,
placebo-controlled comparison of eprodisate to placebo (1:1 randomization)
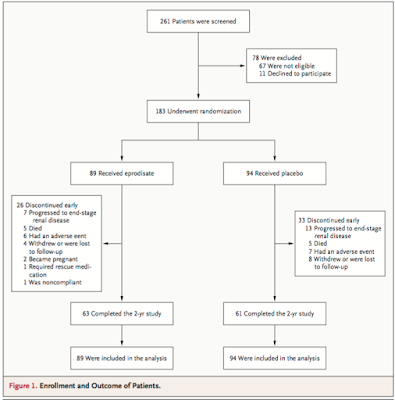
13 countries, 27 centers, 261 patients
screened and 183 enrolled (7/11/2001-2/14/2003)
Key Inclusion criteria :
- Tissue diagnosed AA amyloidosis by histologic demonstration of Congo red staining and birefringence with the use of polarized microscopy and reactivity with anti-AA antibodies by IHC
- Kidney involvement (24hr urine protein >1g x 2 OR CrCl <60ml/min x 2)
Key Exclusion
criteria:
- Kidney disease other than AA amyloidosis,
- Severe renal disease: CrCl <20ml/min OR SCr >3, DM
- Abnormal liver function: LFTs or alk phos >5 xULN, t bili >1.5xULN
- Diabetes Mellitus
- Nephrotic syndrome (Y/N)
- Treatment center
Treatment: 800-2400 mg (depending on renal function) eprodisate/placebo in two divided doses each day for 24 months. Alternating office visits and telephone follow-up calls every two months.
Primary Endpoint:
- Composite endpoint of worsening renal function (SCr >2x BL, CrCL <0.5 BL, or progression to dialysis-requiring ESRD) or death
Key Secondary Endpoints:
- Slope of decline in CrCl over time
- Change in proteinuria
- Change in amyloid quantitation in abdominal fat
- Resolution or development of chronic diarrhea
Results:
- Primary composite endpoint: 42% reduction in the risk of worsening renal function or death (HR 0.58 [95% CI 0.37 - 0.93; p=0.02]). Teasing this apart: renal function worsened in 27% of eprodisate-treated patients vs 40% placebo-treated pts (p=0.06). There was no difference in mortality (HR 0.95 [95% CI 0.27 - 3.29; p=0.94]).
- Secondary endpoints: Mean slope (+/- SE) of change in CrCl (in ml/min/1.73m2) was -10.9(+/-5.1) for the eprodisate grp versus -15.6(+/- 4.1) in the placebo grp (p=0.02), though no statistical difference in risk of progressing to ESRD (E: 7 pts, P: 13 pts, HR 0.54 ([95% CI 0.22 - 1.37; p=0.20]). Also, no difference in change in urine protein loss, change in abdominal fat amyloid content, or incidence of developing chronic diarrhea.
- Safety: Similar rates of AEs and SAEs between grps
- "Sample size...substantial for the rare disease," but not large enough to be powered to assess differences in lower frequency events like progression to ESRD or death
- Finite versus indefinite therapy may have mattered
- PK analysis could have been informative, since more than half of eprodisate pts received modified doses of medication (authors posited that pts with less severe BL renal disease might have been more likely to benefit)
- Modest benefit may be in part due to mechanism of action: SAA oligomers and protofibril levels not reduced (predictably, since blocking SAA-GAG interaction should only affect mature AA fibril formation). Also, persisting albuminuria itself is nephrotoxic (as explained in an old post about kidney injury in AL amyloidosis) and therapy did not reduce proteinuria.
Subscribe to:
Posts (Atom)